|
11/11/2023 0 Comments Electrolyte anode and cathode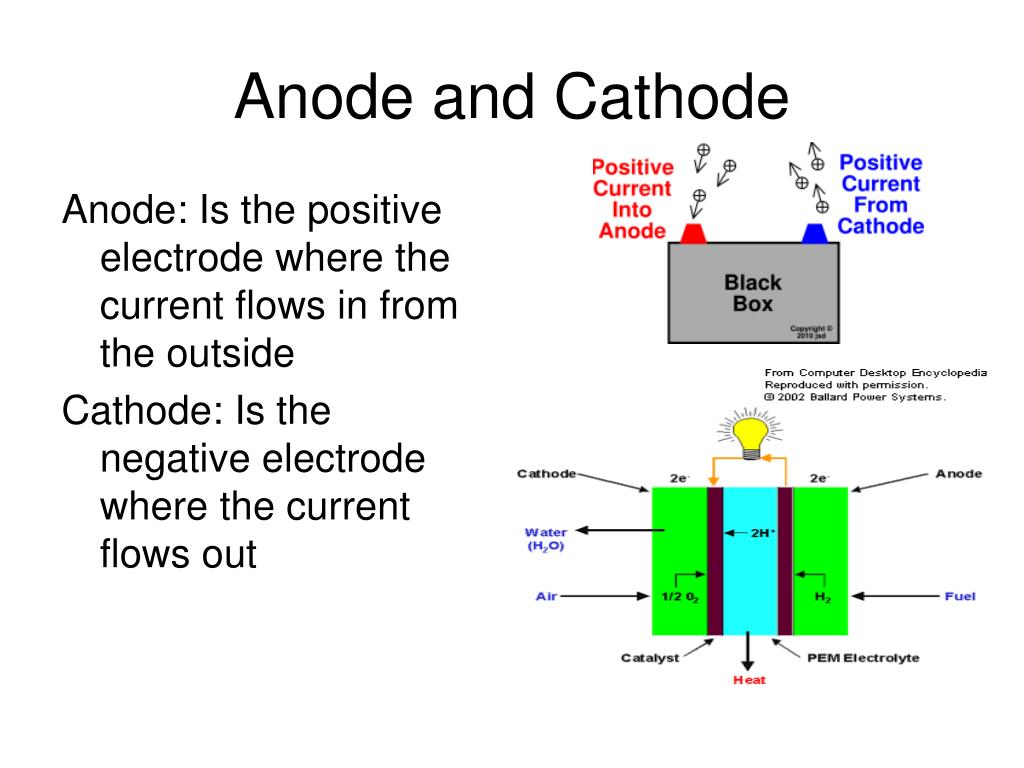
Describe the electrode products, using inert electrodes, in the electrolysis of:.The anions move towards the anode, where they will lose electrons at the anode to become atoms (in a negative ion, there are more electrons than protons, so in order to become an atom, they need to lose those extra electrons to become neutral). The cations move towards the cathode, where they will gain electrons from the cathode to become atoms (in a positive ion, there are more protons than electrons, so in order to become an atom, they need to gain those extra electrons to become neutral). A reaction where electrons are lost is called oxidation and a reaction where electrons are gained is called a reduction. When the ions reach their respective electrodes, they gain or lose electrons. When an electric current is applied, the positive ions move towards the negative electrode, the cathode- this is why the positive ions are called cations and the negative ions move towards positive electrode, the anode- these negative ions are then called anions. In order for a substance to be an electrolyte, it must contain an ionic compound, and the ionic compound must either be molten or dissolved in an aqueous solution. Describe electrolysis in terms of the ions present and the reactions at the electrodes.The positive electrode is called the anode and the negative electrode is called the cathode. These rods are usually inert, as in they do not react- usually graphite or platinum. 
Electrodes are the rods that carry the electric current to and from the electrolyte. The electrolyte is the compound that conducts electricity when molten and breaks down during electrolysis. Use the terms electrode, electrolyte, anode and cathode.State that electrolysis is the chemical effect of electricity on ionic compounds, causing them to break up into simpler substances, usually elements.We apologise for the inconvenience, but hope that the new images will provide you with an even better learning experience. Disclaimer: Due to unforeseen difficulties, we have had to take down the images on this notes page.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
